医脉通编译整理,转载请务必注明出处。
在即将到来的ASCO会议上,计划于芝加哥当地时间6月1日下午进行的乳腺癌口头报告专场,将公布2期研究MDV3100-11的结果。该研究评估了雄激素受体抑制剂恩杂鲁胺用于晚期AR+三阴性乳腺癌的疗效(摘要号1003)。医脉通对此进行了报道。
雄激素受体可能是雄激素驱动三阴性乳腺癌患者的治疗靶点。恩杂鲁胺(ENZA)受体抑制剂,被批准用于转移性去势抵抗性前列腺癌(mCRPC)的治疗。它与比卡鲁胺相比,可以改善转移性去势抵抗性前列腺癌患者的中位PFS (15.7 vs 5.8个月 HR 0.44; p<0.0001)。
MDV3100-11是一项开放标签的、评估单药恩杂鲁胺用于晚期雄激素受体阳性的三阴性乳腺癌患者疗效的2期研究(AR >0% by IHC; NCT01889238)。患者在入组前接受了AR筛查,可存在不可测定的骨转移及之前不限次治疗方案。
研究排除了有中枢神经系统转移或有癫痫史的患者。研究的首要终点为第16周的可评估患者的临床收益(CR, PR or SD)。可评估的患者定义为AR IHC ≥10%,同时产生缓解。研究评估了CBR24,PFS,及安全性。雄激素驱动基因标记(Dx) 根据基因测序和评估结果生成。
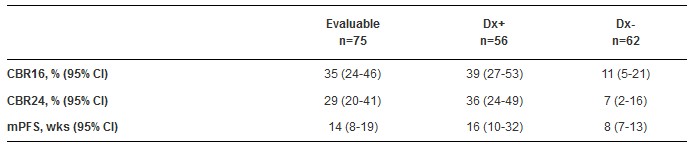
截止至2015年1月16日,404例患者检测了AR IHC。79%的患者的AR>0%; 55%的患者的AR≥10%。18位患者接受了ENZA治疗,43位患者不可评估(29 AR <10%;14 AR ≥10%,但是没有缓解评估)。明确人群的重要结果如下表所示。超过50%的患者接受ENZA作为一线或二线治疗;这些患者中,Dx+患者的mPFS为32周,Dx-患者的mPFS为9周。研究观察到了两例完全缓解,5例部分患者。118位患者中≥10%的患者发生了不良反应事件,包括疲劳(34%),恶心(25%),食欲减退(13%),腹泻及潮热(10%)。疲劳是唯一发生在5%及以上患者中的3度及以上的不良事件。
这是AR抑制剂用于三阴性乳腺癌的最大规模的研究。IHC结果显示,AR的普遍性高于之前的报道。47%的患者有雄激素相关基因标记(Dx+),似乎该组患者的临床结果更好。ENZA的不良反应事件与之前的数据一致。ENZA可能代表了三阴性乳腺癌患者的治疗新选择。此前这类患者只能接受细胞毒化疗药物。临床信息:NCT01889238
摘要原文
Background: The AR may be a therapeutic target for pts with androgen-driven TNBC. ENZA, a potent AR inhibitor, is approved in men with metastatic castration-resistant prostate cancer (mCRPC) and improves median PFS compared to bicalutamide in men with mCRPC (15.7 vs 5.8 mos; HR 0.44; p<0.0001).
Methods: MDV3100-11 was an open-label, Simon 2-stage study evaluating single agent ENZA in advanced AR+ TNBC (AR >0% by IHC; NCT01889238). Pts could be prescreened for AR, and have non-measurable bone disease and unlimited prior regimens; CNS metastases or seizure history were exclusionary. The primary endpoint was clinical benefit (CR, PR or SD) at 16 wks (CBR16) in ‘Evaluable’ pts defined as having both AR IHC ≥10% and a response assessment. CBR24, PFS, response rate, and safety were assessed. An androgen-driven gene signature (Dx) was created from gene profiling and outcomes were assessed accordingly. Stage 2 enrolled if CBR16 was ≥3 of 26 Evaluable pts; H0 was rejected if CBR16 was ≥9 in 62 yielding 85% power at 5% significance to test against a 1-sided alternative (CBR16 ≥20%).
Results: As of 16 JAN 2015, 404 samples were tested for AR IHC: 79% had AR >0%; 55% had AR ≥10%. 118 pts were treated with ENZA; 43 pts were not Evaluable (29 AR <10%; 14 AR ≥10% but no response assessment). Key outcomes in the defined populations are below as shown in the Table. Over 50% received ENZA as 1st or 2nd line; mPFS in these pts was 32 wks in Dx+ and 9 wks in Dx-. Two CRs and 5 PRs have been observed. Related AEs in ≥10% of 118 pts were fatigue (34%), nausea (25%), decreased appetite (13%), diarrhea and hot flush (10%). Fatigue (5%) was the only AE ≥ Grade 3 in ≥5%.
Conclusions: This is the largest study of an AR inhibitor in TNBC. IHC results suggest AR prevalence is higher than previously reported. 47% of pts had an androgen-related gene signature (Dx+) and clinical outcomes appeared superior in this group. AEs from ENZA were consistent with its known profile. ENZA may represent a novel therapeutic option in pts with TNBC who would otherwise receive cytotoxic chemotherapy. Clinical trial information: NCT01889238
更多ASCO精彩内容》》》2015年ASCO年会专题报道
